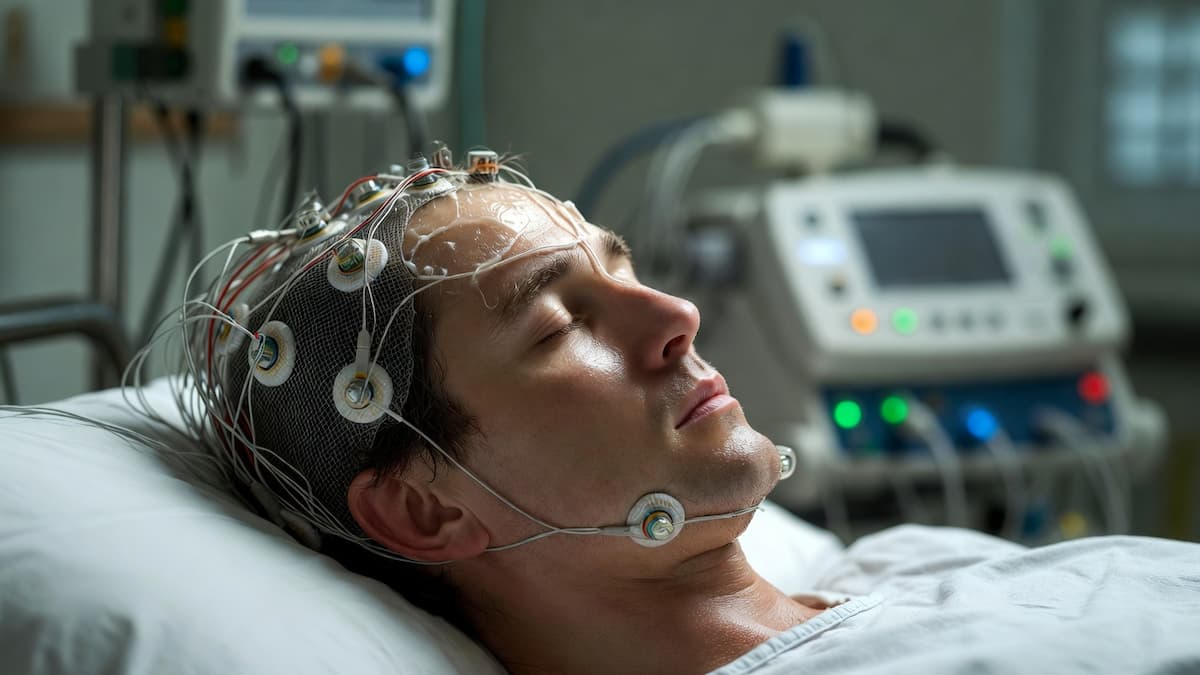
In June 2025, five people with severe paralysis were using Neuralink implants to control digital devices with their thoughts. Not in a laboratory. Not as part of a carefully controlled experiment where every moment was monitored. In their lives — using brain signals to send messages, browse the internet, control wheelchairs.
This is a sentence that would have sounded like science fiction a decade ago. It is now a documented clinical reality.
Brain-computer interfaces began as a technology for control — ways for the brain to communicate with machines. But the most consequential application emerging from BCI research is not control. It is medicine. The same technology that allows a paralyzed person to move a cursor on a screen is revealing itself as a treatment for conditions that have resisted every other approach: drug-resistant epilepsy, severe depression, locked-in syndrome, the silence that descends when ALS takes away the ability to speak.
What a BCI Actually Does in a Medical Context
A brain-computer interface is any system that creates a direct communication pathway between neural activity and an external device. In medical contexts, this means reading electrical signals from neurons — either from the surface of the brain or directly from brain tissue — interpreting them, and using that interpretation to either control something external or to deliver targeted stimulation back into the brain.
Medical BCIs fall into two broad categories.
Active BCIs require intentional user input. A patient with paralysis consciously attempts to move their hand; electrodes detect the motor cortex activity this generates; algorithms translate it into a command — move the cursor left, type the letter A, advance the wheelchair forward. The device amplifies and redirects an intention that the patient’s body can no longer execute.
Passive BCIs operate without conscious effort. These systems continuously monitor brain activity to detect states — the onset of a seizure, the neural signature of depression, the patterns associated with fatigue or pain — and respond automatically. The patient does not need to do anything; the device watches, interprets, and intervenes.
Both types are being developed for therapeutic use, and they address fundamentally different problems.
Paralysis and ALS: Giving Back the Ability to Communicate
For people with complete paralysis — spinal cord injuries, advanced ALS (amyotrophic lateral sclerosis), locked-in syndrome — the most immediate catastrophe is often not the loss of movement itself. It is the loss of communication. The ability to speak, to type, to signal to the people around you that you are present and thinking and have something to say.
BCI research has made remarkable progress on this specific problem.
By June 2025, five individuals with severe paralysis were using Neuralink’s implant to control digital devices with their thoughts. Synchron’s Stentrode — delivered via blood vessel rather than open brain surgery, threaded through the jugular vein to sit in the motor cortex’s draining vein — has allowed patients to send texts, email, and browse the internet using neural signals alone.
In the United States, an estimated 5.4 million people live with paralysis that impairs their ability to use computers or communicate. For even a fraction of these individuals, a working BCI does not just improve quality of life — it reconnects them to the world entirely.
The technology for speech decoding has advanced particularly fast. BCI systems can now decode attempted speech — the motor cortex activity that would have produced spoken words, even when no muscles respond — with sufficient accuracy to reconstruct words and sentences in real time. For a person with ALS who has lost all voluntary movement, this is not an assistive technology. It is the difference between being able to communicate at all and being locked inside a body that cannot respond.
Epilepsy: Predicting the Storm Before It Arrives
Epilepsy affects an estimated 5 million new patients annually worldwide. For most, medication controls seizures adequately. For roughly one-third, it does not — a condition called drug-resistant epilepsy, where seizures continue despite optimal treatment. For these patients, the unpredictability of seizures is itself a major source of disability: they cannot drive, cannot work in certain environments, cannot swim or cook alone.
BCI technology is approaching this problem from two directions.
The first is prediction. Seizures are not entirely random — they are preceded by detectable changes in neural electrical activity, often minutes before clinical symptoms appear. Machine learning algorithms trained on neural data from implanted electrodes can identify these precursor patterns with increasing reliability, providing warnings before a seizure begins.
The second is closed-loop intervention. Once a system can detect the early neural signature of a seizure, it can respond automatically — delivering targeted electrical stimulation to disrupt the seizure’s spread, or triggering the release of medication directly at the seizure focus. This is not a drug. It is real-time neural intervention triggered by the brain’s own activity.
Columbia University researchers developed BISC — the Biological Interface System to Cortex — a single silicon chip with 65,536 electrodes that can be inserted through a small opening in the skull. This level of electrode density means unprecedented resolution in reading neural activity, opening the possibility of detecting and intervening in seizure onset with a precision that existing technology cannot match. BISC is being specifically developed for drug-resistant epilepsy, with NIH funding for clinical trials.
Stroke Rehabilitation: Teaching the Brain to Rewire Itself
Stroke is one of the leading causes of disability worldwide. When a stroke damages motor areas of the brain, patients often lose control of one side of their body — an arm, a hand, a leg. Traditional rehabilitation tries to rebuild function through repetitive physical therapy, exploiting the brain’s neuroplasticity: its ability to reorganize, forming new neural pathways to compensate for damaged ones.
BCIs are making this process faster and more effective.
The key insight is that neuroplasticity is enhanced when motor effort coincides with feedback. When a stroke patient attempts to move their paralyzed hand and simultaneously receives sensory feedback — electrical stimulation of the hand, or visual feedback of a robotic hand moving in response to their neural intention — the brain reinforces the attempted motor pathway more strongly than with physical therapy alone.
BCI-based post-stroke motor rehabilitation shows a medium to large effect size in improving upper limb motor function across multiple randomized controlled trials. Combining BCI training with goal-directed physical therapy has been found to significantly improve hand and arm movements in chronic stroke patients who initially had complete paralysis.
The mechanism is neuroplasticity itself: the BCI creates a closed loop between motor intention and sensory consequence, exactly the kind of reinforcement learning signal the brain uses to form motor skills. By amplifying and delivering this signal precisely when the patient makes the effort, BCIs accelerate the rewiring that rehabilitation aims for.
Depression and Psychiatric Conditions: The Hardest Frontier
The most ambitious — and most contested — medical BCI application is in psychiatry.
Treatment-resistant depression affects tens of millions of people for whom medication, therapy, and even electroconvulsive therapy have failed. Deep brain stimulation (DBS) — delivering continuous electrical stimulation to specific brain circuits through implanted electrodes — has shown effectiveness for some patients with severe depression, but the approach has been inconsistent. Different patients respond to stimulation of different regions; the same stimulation parameters that help one patient may not help another.
The emerging approach uses passive BCIs to make stimulation adaptive. Rather than delivering constant stimulation regardless of the patient’s mental state, a closed-loop system monitors neural biomarkers in real time — patterns of activity associated with depressive states versus more neutral ones — and delivers stimulation only when and where it is needed.
A landmark study at UCSF demonstrated that this personalized closed-loop approach produced dramatic improvement in a patient with severe treatment-resistant depression, with near-instantaneous mood improvement triggered automatically by detection of the patient’s specific neural biomarker. The approach is still experimental and the evidence base is small, but the principle — that psychiatric symptoms have neural signatures that can be detected and intervened upon — has significant implications.
BCIs are being investigated for ADHD, anxiety, PTSD, and Parkinson’s disease. In Parkinson’s, adaptive DBS systems are already in clinical use, delivering stimulation that adjusts in real time based on the patient’s movement state rather than running at constant parameters — a significant improvement over first-generation DBS that reduces side effects while maintaining benefit.
The Hardware Getting Smaller and Less Invasive
One of the practical barriers to BCI medicine has always been surgery. A device that requires opening the skull carries real risks — infection, bleeding, the normal hazards of any brain surgery. Patient populations who might benefit most from BCIs often have comorbidities that increase surgical risk.
The trend in BCI hardware is toward smaller, less invasive, and higher resolution simultaneously.
Synchron’s Stentrode avoids open brain surgery entirely — delivered through blood vessels, it requires no craniotomy. Precision Neuroscience is developing an ultra-thin electrode array designed to slip between the skull and the brain through a small incision. BISC, at Columbia, packs 65,536 electrodes onto a chip thin as a human hair.
The pattern is familiar from the history of other medical devices: pacemakers went from room-sized machines to devices smaller than a coin. Cochlear implants, once elaborate external systems, have become integrated and miniaturized. BCIs are following the same trajectory, driven by the same semiconductor technology.
The next generation of devices is also increasingly wireless. Earlier BCIs required wired connections through the skull — a persistent infection risk and a severe practical limitation. Modern designs use wireless transmission, allowing implanted devices to communicate through the skull without any external penetration.
What This Means for Medicine
The convergence of BCI hardware improvements, AI-based neural decoding, and growing clinical evidence is moving these devices from last-resort experimental treatments toward mainstream neurological care.
In 2025, clinical trials for BCIs have expanded from single-digit to dozens of patients. Companies like Neuralink and Synchron are beginning trials in countries outside the United States. The FDA has granted breakthrough device designations to multiple BCI products, a pathway that accelerates review for devices that may provide more effective treatment for serious conditions.
The BCI healthcare market, projected at over $160 billion in 2024 by some estimates, is growing at 10-17% annually — driven not by consumer technology but by clinical need. Paralysis, epilepsy, stroke, depression, ALS: these are the conditions drawing investment, and they are conditions that affect hundreds of millions of people worldwide who currently have inadequate treatment options.
What is emerging is a new category of medicine — one that treats disease not by correcting chemistry with drugs, but by correcting computation with electronics. The brain runs on electrical signals. BCIs speak that language directly.
The question that remains is not whether this technology will become part of medicine. It already is. The question is how quickly the evidence base will grow, how accessible it will become, and whether the healthcare systems, insurers, and regulatory frameworks that govern medicine can keep pace with technology that is advancing faster than any of them anticipated.
For the five people using Neuralink to communicate with their thoughts, that question is already answered. The medicine is working. The rest of the world is catching up.